Intramolecular transesterification of depsides yields fluorescent 1H-isochromen-1-ones: Application as a chemical probe for lichen determination
Résumé
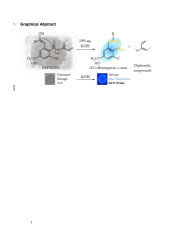
The reactivity of eight purified depsides obtained from six european lichens and that display as 2-oxoalkyl chain in ortho-position of the ester bond was explored. These depsides were found to lead to 1H-Isochromen-1-ones, which exhibit a distinctive blue fluorescence at 365 nm, in the presence of a 10% aqueous solution of KOH. A mechanistic explanation, involving the formation of an enolate intermediate and intramolecular transesterification, was proposed and validated by DFT. By exploiting this fluorescent phenomenon, we conceived a chemical probe (the KUV probe) that is useful for lichen determination, as exemplified on a selection of European Porpidia species.
Fichier principal
 MS_Ferron_et_al_2021_minor_revisions_without_highlights.pdf (1.08 Mo)
Télécharger le fichier
MS_Ferron_et_al_2021_minor_revisions_without_highlights.pdf (1.08 Mo)
Télécharger le fichier
Origine : Fichiers produits par l'(les) auteur(s)
